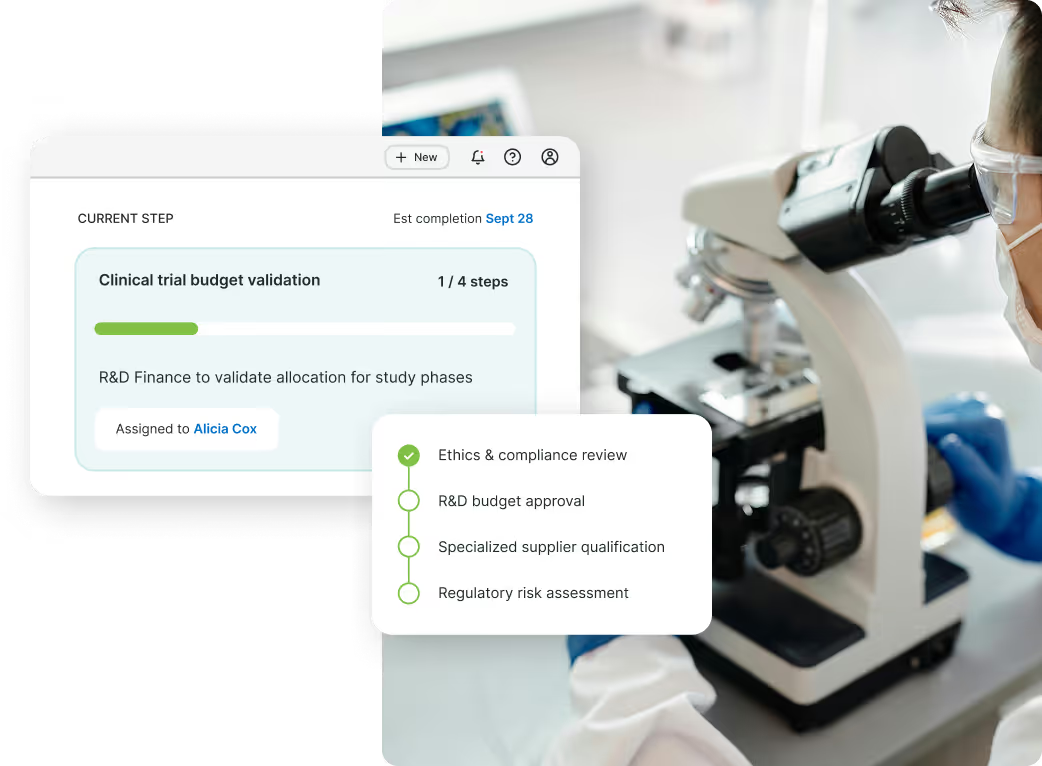
How does ORO support procurement and compliance in life sciences organizations?
ORO helps life sciences companies automate and orchestrate complex procurement, supplier management, and compliance workflows across clinical, R&D, manufacturing, and corporate operations. The platform connects procurement, QA, regulatory, legal, finance, and IT teams into a unified workflow experience that improves speed, traceability, and governance across highly regulated environments.



















.avif)

.svg)



.avif)

.svg)


